AOD9604 vs Semaglutide: Comparing the Most Talked-About Anti-Obesity Drugs

Obesity and metabolic disorders are on the rise worldwide, driving demand for effective pharmacological solutions that support weight management and improve overall health. Among the compounds gaining attention in recent research, AOD9604 and semaglutide stand out as two of the most promising—but fundamentally different—anti-obesity drugs.
In this article, we explore their mechanisms of action, clinical evidence, safety profiles, and real-world data, providing an in-depth comparison to help researchers, healthcare professionals, and patients make informed decisions.
What Are AOD9604 and Semaglutide?
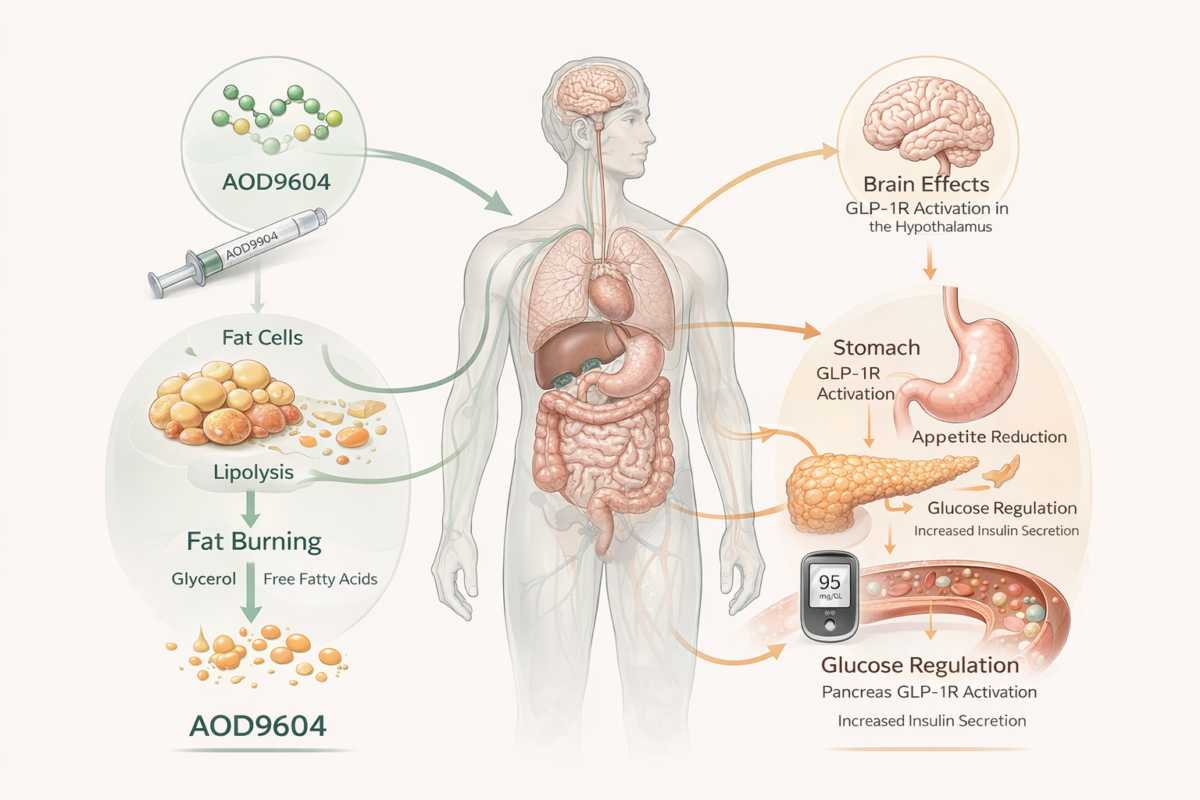
AOD9604 is a synthetic peptide fragment derived from human growth hormone (hGH). Designed to preserve hGH’s fat-burning (lipolytic) properties while avoiding its growth-promoting effects, it remains largely experimental.
Semaglutide, on the other hand, is a fully approved GLP-1 receptor agonist. It is widely recognized for managing type 2 diabetes and producing clinically significant weight loss. Unlike AOD9604, semaglutide has strong regulatory backing, extensive clinical trials, and a growing body of real-world evidence supporting its safety and effectiveness.
How Do They Work? Mechanisms of Action
AOD9604: Targeted Fat-Burning Peptide
AOD9604 is a modified fragment of hGH (amino acids 176–191) designed to focus on fat metabolism. Preclinical studies suggest it may:
- Promote lipolysis (breakdown of stored fat)
- Inhibit adipogenesis (formation of new fat cells)
- Modulate metabolic signaling pathways downstream of hGH
However, the exact mechanism is not fully understood, and AOD9604 has not yet been approved for obesity treatment. Its effects are still under investigation in experimental and off-label contexts.
Semaglutide: Appetite Suppression and Metabolic Control
Semaglutide mimics the incretin hormone GLP-1 to deliver multiple metabolic benefits:
- Increases insulin secretion in response to glucose
- Suppresses glucagon release, stabilizing blood sugar
- Slows gastric emptying, reducing appetite
- Directly acts on brain regions that control hunger
These combined effects result in significant, sustainable weight loss and improved glucose control for patients with type 2 diabetes. Semaglutide’s mechanism is well-established and backed by numerous clinical trials.
Clinical Evidence: Semaglutide Leads the Way
There is a stark contrast in clinical data between the two drugs.
Semaglutide:
- Proven effective in multiple large-scale randomized controlled trials (RCTs), including the PIONEER and SUSTAIN programs
- Reduces body weight and improves cardiovascular risk factors
- Demonstrates efficacy across diverse populations and geographies
- Supported by meta-analyses and real-world studies
AOD9604:
- Limited human trial data
- Mixed results in weight loss outcomes
- Primarily experimental, with no regulatory approval
- Lack of large, well-controlled studies limits clinical confidence

Safety and Side Effects
Semaglutide
Semaglutide’s safety profile is well-documented. Common side effects include nausea, vomiting, and diarrhea. Beyond clinical trials, digital pharmacovigilance—using social media and real-world patient reports—has revealed additional insights, including rare but serious events like pancreatitis.
AOD9604
AOD9604’s safety profile remains largely unknown due to limited clinical exposure. Available data suggest it does not cause the growth-promoting or glucose-altering effects seen with full-length hGH. However, the absence of systematic post-market surveillance means its risk profile is uncertain.

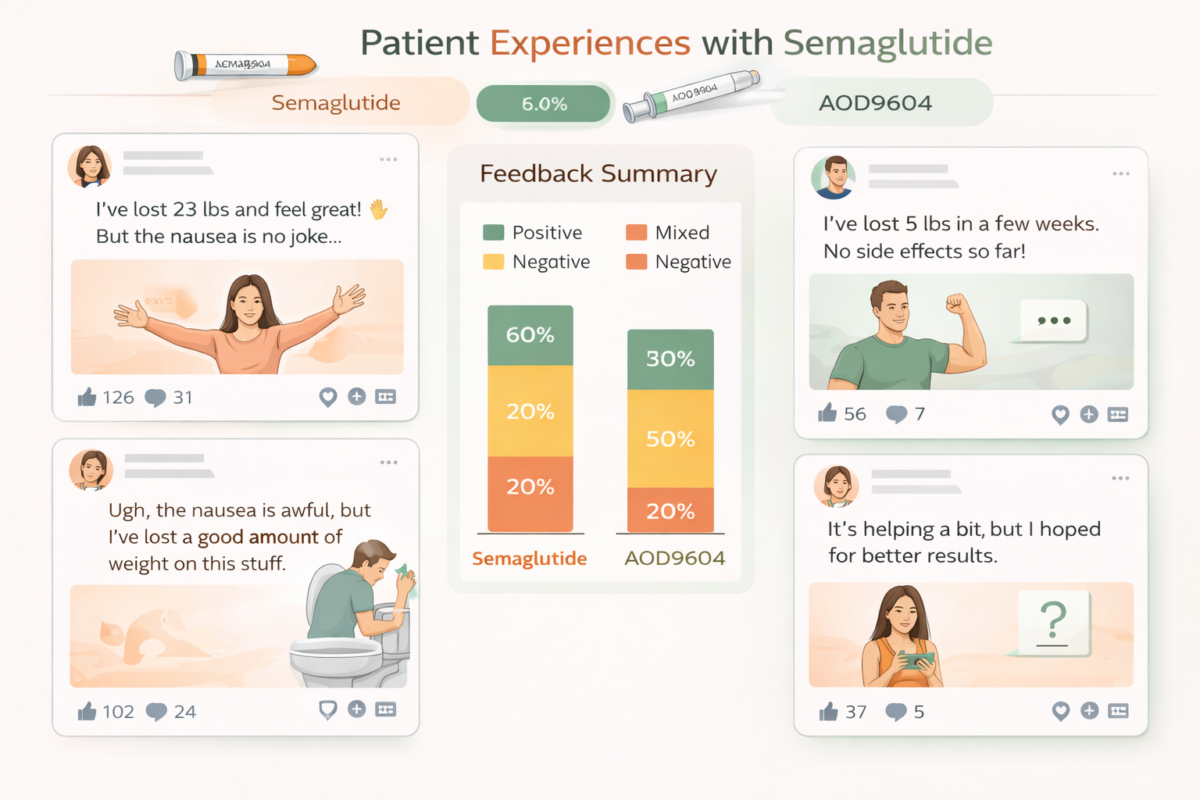
Real-World Insights: Patient Experiences Matter
Real-world data, including social media sentiment analysis, has become invaluable in understanding drug effectiveness and patient satisfaction.
-
Semaglutide: Over 850,000 social media posts reveal patient experiences, side effects, and concerns about cost and accessibility. This data complements clinical trials to inform patient-centered care.
-
AOD9604: Minimal online presence and patient reports limit insights into real-world efficacy and safety.
Key Takeaways for Healthcare and Research
The comparison of AOD9604 and semaglutide highlights several lessons:
-
Semaglutide exemplifies evidence-based anti-obesity therapy, combining clinical validation, regulatory approval, and patient feedback.
-
AOD9604 demonstrates the risks of using experimental compounds without robust clinical and regulatory oversight.
-
Integrating clinical trial data with real-world patient experiences is essential for optimizing obesity treatment outcomes and ensuring safety.
Conclusion: Choosing an Anti-Obesity Therapy
Semaglutide stands out as a well-researched, clinically validated, and real-world-tested anti-obesity medication. Its proven efficacy, clear mechanism, and robust safety profile make it a reliable choice for patients and healthcare professionals.
AOD9604, while mechanistically intriguing, remains largely experimental and lacks the clinical validation and regulatory support necessary for mainstream use.
For effective, safe, and evidence-based weight management, combining rigorous clinical data with real-world insights is the future of obesity treatment and anti-obesity drug research.
